- ≡ Hypnum uncinatum Hedw., Sp. Musc. Frond. 289 (1801)
- ≡ Drepanocladus uncinatus (Hedw.) Warnst., Beih. Bot. Centralbl. 13: 417 (1903)
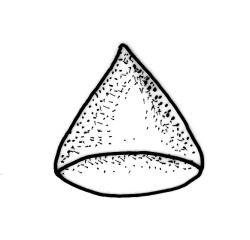
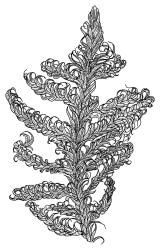
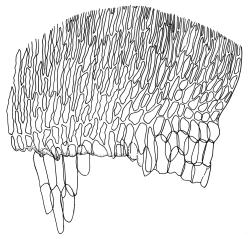
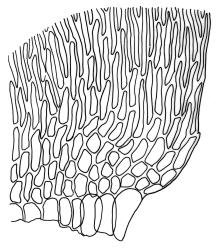
Plants medium-sized to robust, yellow-green, bright-green, or rarely yellow-brown, usually forming dense wefts. Stems creeping or ascendant in self-supporting wefts, commonly 30–80(–100 mm), irregularly or subpinnately branched or occasionally nearly unbranched, in cross-section with a hyaloderm (with cells often stripping off with stem leaves), 4–5 layers of thick-walled cells beneath the hyaloderm, and a small but distinct central strand. Stem leaves strongly falcate-secund to circinate, closely-spaced, mostly c. 3.5–4 mm, ovate-lanceolate, narrowed to a very slender apex, plane at margins and sharply serrulate above, strongly plicate, not or slightly decurrent, not or little altered when dry; upper laminal cells smooth, linear, and flexuose, c. (36–)54–75 × c. 4 µm, firm-walled, not or very weakly porose, scarcely differentiated at apex or at margins; basal laminal cells shorter, thicker-walled, porose, and often pigmented in a few rows at insertion; alar cells inflated and hyaline in groups of c. 8–10 cells which are bounded above by oblong to ± isodiametric cells which grade rapidly into the laminal cells. Costa rather narrow, unbranched and mostly reaching into the base of the acumen, often obscured by plications. Branches variable in length. Branch leaves smaller and less distinctly plicate; the alar group smaller, usually with 2–3 inflated cells in extreme angles. Pseudoparaphyllia present, foliose, mostly broader than long. Paraphyllia absent.
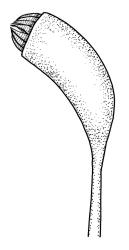
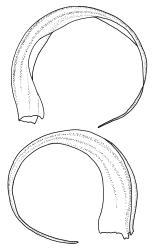
Autoicous. Perichaetia scattered on stems, with the inner leaves sheathing, narrowly lanceolate and finely filiform at apex, strongly plicate, to c. 6 mm. Perigonia scattered on stems. Setae 15–35 mm, red-brown, smooth, straight or weakly flexuose, twisted weakly to right above; capsules oblong-cylindric from a weakly defined and tapering neck, strongly curved, smooth, constricted below the mouth when dry, 1.5–2.0 mm; exothecial cells oblong to nearly round, firm-walled; stomata as per family; annulus strongly differentiated, not revoluble, usually adhering to capsule mouth; operculum high-conic and short apiculate. Exostome teeth yellow-brown, cross-striolate below and papillose above on outer surface, bordered, appendiculate at margins (by laterally projecting trabeculae), strongly trabeculate but otherwise nearly smooth on inner surface; endostome from a high basal membrane, giving rise to narrowly perforate segments equal the teeth in height, with single weakly nodose cilia (in N.Z. material). Calyptra cucullate, smooth. Spores 16–20 µm, finely papillose.
Crum & Anderson 1981, fig. 474 (as Drepanocladus uncinatus); Kanda 1977, fig. 46, 10–23; Seppelt 2004, fig. 50; Meagher & Fuhrer 2003, p. 81; Ochyra et al. 2008, fig. 242.
Difficulty sometimes can occur separating sterile material of Sanionia uncinata from Brachythecium paradoxum. Plants of S. uncinata are more robust with stems leaves that are considerably longer (generally >3.5 mm vs c. 2–3 mm), more plicate, and less toothed than the leaves of B. paradoxum. The moderate differentiation of the stem and branch leaves in S. uncinata gives the plants a different aspect than B. paradoxum. When fertile, the erect and strongly sheathing perichaetial leaves and the smooth setae in the present species contrast sharply with the widely spreading perichaetial leaves and the papillose setae of B. paradoxum. Sanionia uncinata plants invariably have thin-walled and inflated cells in the extreme alar angles while those of B. paradoxum are small and thick-walled. Thin-walled hyalodermis cells nearly always remain attached to stripped-off stem leaves in S. uncinata, while these are absent in the Brachythecium.
NI: S Auckland (Coromandel, Maunga Pōhatu, Ātiamuri), Gisborne (Mt Hikurangi), Hawke’s Bay (Kāweka Range), Taranaki (Mt Egmont), Wellington (Tongariro National Park, Ruahine Range); SI: Nelson, Marlborough (Mt Stokes), Canterbury, Westland (Ōtira, Kelly Range, Croesus Track), Otago, Southland; M. Material from A and C has not been seen but Seppelt (2004) records it from both these localities. While S. uncinata is a very common species in much of South I. (especially east of the Main Divide) it is documented from relatively few localities on North I.
Bipolar. Argentina*, Chile*, South Shetland Is.*, Marion I.*, East Africa*, and widespread in northern hemisphere. Recorded as rare from Tasmania by Dalton et al. (1991); recorded from mainland Australia but considered "not common" there by Scott & Stone (1976, p. 417). Seppelt (2004) cited additional southern hemisphere localities.
Occurring at moderate to high elevations in southern beech montane forest, subalpine scrub and alpine grasslands and herbfields, most commonly and conspicuously east of the Main Divide and in the Otago mountains on SI. Also occurring in a variety of wetter sites including tarn margins and flushes and as an epiphyte in hyper-moist subalpine forest. Apparently restricted to above c. 1000 m on North I and ranging from 700–1830 m (Craigieburn Range, Canterbury L.D.) on SI. When occurring on the forest floor it is often with Dicranoloma spp., Pyrrhobryum mnioides subsp. contortum, and Lepidozia spp. In high-altitude wetlands and flushes associated it is often associated with Aulacomnium palustre, Climacium dendroides, and Sphagnum cristatum as well as other less common species.
N.Z. material appears to have a less distinct central strand than northern hemisphere material. The hyaloderm is sometimes inconspicuous in stem cross-section but some thin-walled cells usually adhere to the base of stripped-off stem leaves. According to Rod Seppelt (pers. comm., 4 April 2013) the outer wall of the hyaloderm cells is thin and collapses with age, leaving the radial walls prominent. This partial collapse causes the stem to appear like a cog-wheel when viewed in cross-section.
Material from high-altitude wetlands is often brown-green to nearly bronze in colour and has stems less branched than usual for the species.