- ≡ Leskea dendroides Hedw., Sp. Musc. Frond. 228 (1801)
- = Climacium novae-seelandiae Müll.Hal. ex Beckett, Trans. & Proc. New Zealand Inst. 25: 292 (1893)
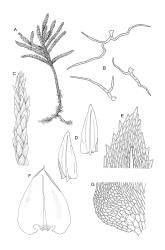
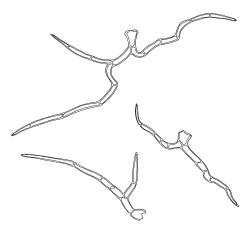
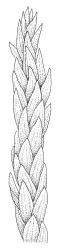
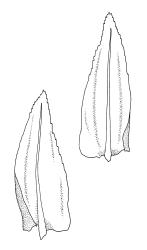
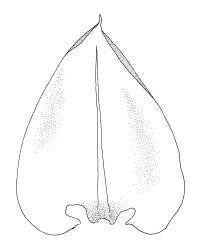
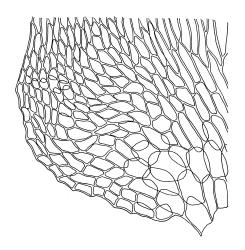
Plants ± glossy when dry, bright green, yellow-green, or rarely bronze. Secondary stems erect and dendroid with a well-differentiated stipe and frond, simple or rarely sparingly branched, mostly 30–70(–100) mm high, red-brown below, densely beset with smooth, much-branched, red-brown rhizoids. Stipe leaves erect-appressed when moist or dry, obscuring the stipe, broadly ovate and cordate, broadly acute or obtuse at apex, sharply mucronate, serrulate near apex, entire below, 2.3–2.8 × c. 2.0 mm. Frond to c. 45 mm diam. Branches variable in length, up to c. 22 mm, mostly unbranched. Branch leaves ovate-lanceolate, c. 1.6–2.2 × 0.8–1.0 mm, coarsely or sometimes weakly serrate in upper half, serrulate or entire below, weakly sulcate, auriculate and scarcely decurrent; upper laminal cells mostly 30–45 µm long, weakly porose; alar cells as per genus. Costa strong, failing c. 5–8 cells below apex, often ending in a single abaxial tooth.
Perichaetial leaves squarrose. Perigonia and capsules not seen in N.Z. material.
Brotherus 1925, fig. 479; Sainsbury 1955, pl. 49, fig. 1; Crum & Anderson 1981, fig. 611; Smith 2004, fig. 234, 1–4.
Because of its frondose habit, C. dendroides could be confused with members of the genus Sciadocladus, particularly S. kerrii. The overall habit of these two plants is different, however, in part because the stipe leaves in C. dendroides are broadly acute to obtuse, mucronate, and erect-appressed, obscuring the stipe in both the dry and moist condition. The stipe leaves of all Sciadocladus spp. are acuminate, widely spreading when moist, and do not obscure the stipe. The branch leaves in C. dendroides are broader apically and have shorter upper laminal cells (c. 30–45 µm), while the branch leaves of S. kerrii are more acute and have longer (mostly 45–95 µm) upper laminal cells. The costae of C. dendroides lack multiple abaxial spines near their apex in contrast to species of Sciadocladus. Climacium dendroides is not known to fruit in N.Z., while species of Sciadocladus frequently fruit. Confusion between the terrestrial C. dendroides and the epiphytic Braithwaitea sulcata seems less likely; the latter differs in many ways including the pinnately branched and flattened nature of its fronds and the presence of filiform axillary gemmae. Confusion with other species in the N.Z. flora is not likely.
NI: Wellington (Upper Makatote River); SI: Nelson, Marlborough (Mt Tapuaenuku), Canterbury, Westland, Otago, Southland (S Arm of Lake Manapōuri, Lake Gunn).
Adventive. Tasmania*, Australia (Victoria*), North America*, Europe*, E Asia*. A literature report from Tasmania was cited by Martin (1946), but this record was doubted by Dalton et al. (1991). However, L. Cave (pers. comm., Sep. 2004) has rediscovered the species in Tasmania.
On humic soil at margins of lakes, tarns, and streams, in seepage slopes, damp depressions in tussock grasslands (especially Chionochloa rubra or C. australis grasslands), and cushion bogs. In cushion bogs it is sometimes dominant atop hummocks. Occasionally in swampy areas or adjacent to watercourses in subalpine Nothofagus forest. This species can also occur abundantly in roadside ditches (as at the Taramakau Valley, Westland L.D.) and occasionally at low elevations on dry roadside gravels (as at Waimangaroa, Westland L.D.). On such dry gravels it can assume confusing irregularly branched and prostrate forms. Bryophyte associates include Aulacomnium palustre, Breutelia pendula, Campyliadelphus polygamum, Sanionia uncinatus, Sphagnum spp., and Syntrichia rubra. The single North I. collection (one stem only) was associated with Hylocomium splendens. On North I. known only from 1900 m elevation. On South I. occurring mostly at c. 470–1600 m, but extending to lower elevations (c. 30 m) in Westland and Southland and upwards to c. 2000 m in the ranges of western Otago (e.g., Remarkable Range).
The earliest confirmed collection was made in 1891 from Castle Hill, Canterbury L.D., by T. Kirk (CHR 510623, type of C. novae-seelandiae Beckett). An unknown collector gathered it in the Rock and Pillar Range in Otago L.D. in 1892 (CHR 623649) and Kirk made an undated but early collection at Lake Rotoiti in Nelson L.D. (CHR 623650). Subsequently, there is an inexplicable collection gap of roughly 40 years until 1931. Since 1950 the species has been collected in nearly all parts of South I. and there are c. 55 gatherings of this species in CHR.
The absence of gatherings by numerous capable South I. collectors in the late 19th and early 20th centuries and the apparent rapid increase in the collection frequency of this species since 1950 strongly suggest that C. dendroides is adventive in N.Z. Given its alleged adventive status, its occurrence in seemingly unmodified native vegetation, sometimes in remote localities (such as at the cirque at the head of the Rahu River in the Victoria Range, Nelson L.D.), is difficult to explain. However, the paucity of pre-1950 collections of such a distinctive and conspicuous species leads to the conclusion that it is a 19th century introduction to the N.Z. flora. The habitat, general distribution, and collection history of C. dendroides exhibit similarities to those of Aulacomnium palustre.
Although extremely variable in stature, the microscopic features of C. dendroides are very constant. Perichaetia are uncommon (seen only in lowland material from Westland L.D.). Shorter and more compact plants are associated with drier habitats.
Horton & Vitt (1976) included N.Z. material in a detailed morphometric study of Climacium dendroides and C. americanum from North America, and concluded that N.Z. material falls within the range of variability of the former species. Dixon (1927, p. 241) reached an identical conclusion.