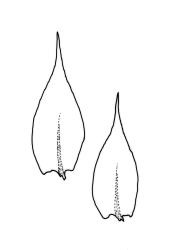
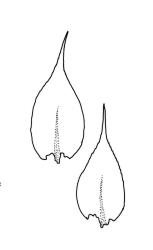
Plants yellow- to dark green, lustrous. Stems irregularly branched, mostly weakly complanate, commonly to c. 20 mm long, in cross-section with central strand apparently absent. Branches mostly c. 5–12 mm, variably complanate (often more complanate in lower segments and scarcely at all near apices; occasionally markedly complanate). Stem and branch leaves differing only by size, not plicate, spreading when moist, little altered when dry. Stem leaves ovate-lanceolate and ± abruptly acuminate, weakly concave, weakly narrowed to the insertion, ± symmetric, neither plicate nor decurrent, (1.1–)1.2–1.5 × 0.4–0.5 mm, finely denticulate nearly to base; mid laminal cells linear-rhombic, mostly (45–)75–90 × c. 6–7 µm and 9–15: 1; cells of the acumen scarcely shorter; cells at insertion shorter, thicker-walled and porose in several rows; alar cells subquadrate in a small and poorly differentiated group. Branch leaves slightly smaller (c. 1.1–1.4 × 0.30–0.55 mm). Costa single, distinctly tapered, extending ½ or more of the leaf length (often to c. ¾ the leaf length and less tapered in stem leaves), often with a terminal abaxial spine.
Autoicous. Perichaetia c. 2 mm long, the leaves erect. Perigonia scattered and usually numerous on fruiting stems, c. 0.6–0.8 mm, nearly black, with filiform paraphyses. Setae flexuose, not twisted, (5)8–15 mm, red-brown, finely and densely papillose throughout (papillae scarcely visible under a hand-lens); capsules short oblong-cylindric, weakly curved, dark brown at maturity, c. 2.0 mm; exothecial cells oblong, firm-walled, distinctly or weakly collenchymatous; operculum c. 1.5 mm, curved, distinctly shorter than the theca. Exostome teeth as per genus, c. 450–500 µm; endostome as per genus, with widely perforate segments and paired or single nodose cilia. Spores 13–15 µm, green.
Wilson 1854, pl. 89, fig. 3; Hedenäs 2002, fig. 11 e.
Sporophytically, the finely scabrose setae and the collenchymatous exothecial cells are distinctive, and, fortunately, collections devoid of sporophytes are relatively uncommon. Differentiation from Eriodon cylindritheca (q.v.) is not always possible when fruit are lacking.
When sterile (or when admixtures occur) this species can be very difficult to differentiate from other species of Rhynchostegium and Eriodon cylindritheca. Rhynchostegium muriculatum and R. laxatum have laminal cells of similar length, but R. muriculatum generally has narrower stem leaves (c. 0.4–0.5 mm wide) than does R. laxatum (c. 0.6–1.0 mm), but this may not consistently distinguish some collections. Rhynchostegium muriculatum has branches that are not or only weakly complanate and mid laminal cells that are generally longer (75–90 µm) in contrast to the decidedly complanate branches and shorter stem leaf mid laminal cells (57–75 µm) of Eriodon.
When fruiting, the scabrose setae (vs smooth in R. laxatum and R. tenuifolium) and the shorter and stouter operculum with a distinctly conic base (vs a slender operculum, which is more or less the length of the theca in R. laxatum and R. tenuifolium) will distinguish R. muriculatum from its N.Z. congeners. However, largely because the papillae in R. muriculatum are low and inconspicuous under the hand-lens or stereo microscope, some herbarium specimens are misnamed as R. laxatum.
Rhynchostegium muriculatum is also sometimes confused with Amblystegium serpens but differs subtly in habit due to the ± complanate set of the leaves in the present species. When fruiting, the scabrose setae, dark brown capsules with collenchymatous exothecial cells, distinctly brown-coloured exostome teeth, and rostrate operculum contrast sharply with the smooth setae, pale brown capsules with non-collenchymatous exothecial cells, pale exostome teeth, and conic operculum of the Amblystegium. The longer (c. 9–15:1) mid laminal cells and weakly differentiated alar group of R. muriculatum also contrast with the shorter (c. 3–5:1 and c. 24–30 µm long) mid laminal cells and more strongly differentiated alar group of the Amblystegium .
NI: N Auckland, including offshore islands (TK, PK, HC, GB), S Auckland, Gisborne, Hawke’s Bay, Wellington; SI: Nelson, Marlborough, Canterbury, Westland, Otago, Southland; St.
Australasian. Tasmania*, mainland Australia*. Reported from Norfolk I. by Streimann (2002).
On wood or bark of both standing trunks and logs in varying stages of decay; on bark of living trees of a wide range of species (including Aristotelia serrata, Carpodetus serratus, Dacrycarpus dacrydioides, Fuchsia excorticata, Veronica elliptica, Melicytus ramiflorus, Myoporum laetum, Pseudopanax sp., Salix sp., and Schefflera digitata, as well as tree-ferns). Also occurring on rock (breccia, basalt, mudstone, greywacke, and granite) or soil, and in both instances often associated with stream beds. Occurring in a wide range of lowland forest and scrub vegetation. On both the North I. and the South I. ranging near sea level to c. 760 m (Tangiwai, Wellington L.D.). Associated with a wide range of bryophytes including Alleniella hymenodonta, Calyptopogon mnioides, Camptochaete pulvinata, Cryphaea spp.,Dichelodontium nitidum, Pyrrhobryum bifarium, Racopilum convolutaceum, Rhynchostegium laxatum, R. tenuifolium, Sauloma tenella, Thuidiopsis sparsa, Metzgeria spp., and Chiloscyphus muricatus.
This is a morphologically variable species. The width and shape of the leaves, the length of the mid laminal cells, the strength of leaf margin toothing, and the degree of flattening of the shoot (both stems and branches) all vary markedly here. Like Scott & Stone (1976, p. 426), I find that a terminal costal spine is often present in this species and strongly doubt Sainsbury’s (1955, p. 436) statement that the nerve here is “never ending in a dorsal spicule”. However, the observational difference may in part be a function of intra-specific variability. The usually present costal spine is often most easily observed when an intact branch is observed under the compound microscope. Forms with broader leaves occur that, if sterile, cannot be differentiated from R. laxatum. In difficult material the shorter stems and lack of leaf striolations in the present species can allow at least tentative determinations. Occasionally material of R. muriculatum is seen in which the laminal cells are quite short (c. 45–60 µm). Molecular investigations might cast some light on the nature of the intra-specific variation. For the present, however, it is advisable to take a wide view of this species.
The type material of R. novae-zealandiae Dixon is very attenuate, but exhibits no morphological differences (including the degree of differentiation of alar cells) to justify its segregation. The presence of Catagonium nitens mixed with one isotype suggests its collection from a very sheltered and shaded site.
Only one sterile specimen from Norfolk I. has been examined, and its identity is not certain. It has well-developed stem and branch leaves that are shorter (c. 0.8–1.2 mm), more ovate, and broader at the apex than representative N.Z. R. muriculatum. However, Streimann’s (2002, p. 12) description suggests that at least some Norfolk I. material is correctly referred to this species.