- ≡ Isothecium pulvinatum Hook.f. & Wilson in Wilson, Bot. Antarct. Voy. II (Fl. Nov.-Zel.) Part II, 105 (1854)
- ≡ Ptilocladus pulvinatus (Hook.f. & Wilson) Lindb. in Paris, Index. Bryol. 234 (1894) nom. inval.
- ≡ Thamniella pulvinata (Hook.f. & Wilson) Besch. ex Paris, Index. Bryol. 234 (1894) nom. inval.
- = Camptochaete beckettii Broth., Öfvers. Finska Vetensk.-Soc. Förh. 42: 114 (1900)
- = Camptochaete falcifolia Broth., Bernice P. Bishop Mus. Bull. 40: 23 (1927)
- = Stereodon maculosus Dixon, Bull. New Zealand Inst. 3: 337 (1929)
- ≡ Camptochaete maculosa (Dixon) Sainsbury, Rev. Bryol. Lichénol., n.s. 21: 222 (1952)
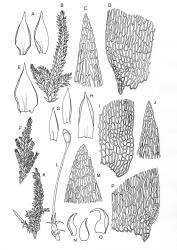
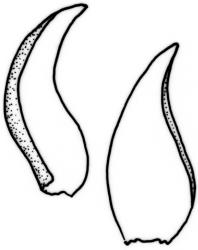
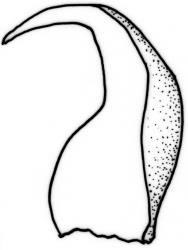
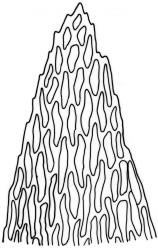
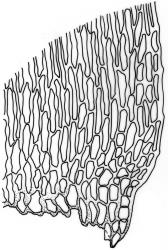
Plants slender to robust, (10–) 15–25 (–40–50) mm, dark to pale, green or green-brown, or olive green. Stems ± erect to scrambling, moderately complanate, stipitate; in cross-section oval, 530 × 420 µm with an outer layer of 3–4 cells surrounding a parenchyma and an indistinct central strand. Stipes often weakly developed, 4–7 mm. Fronds loosely and ± irregularly (bi-) pinnate, complanate. Branches straight, distichous, moderately complanate. Frond axis leaves falcate-secund, not altered when dry, concave, oblong-ovate, acute to finely acuminate, entire, (1.4–) 1.6–2.0 × 0.5–0.6 mm, with alar cells forming an often extensive group 10 cells wide and extending c. 14 cells from the leaf base. Branch leaves smaller, sometimes denticulate to the lower margin, with a smaller alar group, 1.0–1.2 (–1.6) × 0.3–0.5 mm. Costa double and failing below mid leaf, faint, sometimes absent. Mid laminal cells not porose, (40–) 53–58 (–85) × 5 µm; becoming moderately porose below to form a weakly differentiated basal band, (18–) 30–50 × 8–10 µm; those above 32–40 × 5 µm, at the extreme apex c. 12–18 (–22) × 5–8 µm.
Dioicous or pseudautoicous. Setae 8–10 mm. Capsules inclined to horizontal, c. 2 mm long; exothecial cells mostly 28–48 × 20–28 µm. Operculum conic, blunt or apiculate, c. 0.7– 0.8 mm long. Exostome teeth c. 500 µm; endostome with a basal membrance ½ the exostome. Calyptra c. 2 mm. Spores 10–12 µm.
Tangney 1997, fig. 24.
C. pulvinata forms low often dense wefts on rocks and the lower bark of trees. It is easily distinguished by its falcate-secund leaves. It is sometimes difficult to separate from some forms of C. deflexa which may rarely have falcate leaves, but the latter usually lacks the numerous alar cells of C. pulvinata.
While the falcate-secund leaves are unusual in Camptochaete, the rigid stems, dendroid-stipitate growth form and the laminal cell pattern are typical. The sporophyte is that of the genus
C. pulvinata exhibits two forms: a larger (25–40 (–50+) mm) southern form with widely acute leaves and perichaetia and an often small (c. 5–7 × 5–7) group of alar cells, and a northern form which is smaller (10–15 (–20) mm), and has narrower leaf apices, aciculate perichaetia and numerous alar cells (often forming a group 14 × 10–12). However, because of variation within both forms, they are not well separated morphologically and are not here formally recognised. Beever (1986, 1990) has noted the variability of leaf and cell form in C. pulvinata and she treated C. maculosa (Dixon) Sainsbury as a synonym of the former. The features of C. maculosa are characteristic of the northern form.
NI: N Auckland, including offshore islands (TK, HC, GB, LB, Whale I.), S Auckland, Gisborne, Taranaki (Taranaki Maunga), Wellington; SI: Nelson, Marlborough, Canterbury, Westland, Otago, Southland.
Endemic? Also recorded for Hawaiʻi *, see note below.
Common in lowland forests on rocks and the lower bark of trees, it also extends to mid elevations. It occurs in a wide range of mostly shaded habitats from wet forest near streams in Fiordland to drier habitats such as rocks under coastal kanuka scrub. Altitudinal range: near sea level (Akatore Creek, Otago LD) to 940 m (Mt Cook National Park) and 1000 m on Mt Fyffe (Marborough LD) on the South I., and near sea level on Little Barrier Island, Great Barrier Island, and Raukokore River (Gisborne LD) to 1310 m (north end of Kaweka Range, Hawke’s Bay LD) on the North I.
Camptochaete falcifolia Broth. from Hawaiʻi is treated here as a synonym of C. pulvinata. Bartram (1933) compared the type of C. falcifolia with C. pulvinata and found it to be ‘almost in complete accord' with C. pulvinata, and both Hoe (1974) and Miller et al. (1978) followed Bartram in accepting the synonomy. Camptochaete falcifolia is apparently only known from the type specimen. This specimen is incomplete, lacking sufficient material to establish whether or not the plants are stipitate, but it is otherwise typical of C. pulvinata in its wiry pinnate fronds and falcate weakly costate leaves (Bartram 1933; Tangney 1997). Camptochaete pulvinata is otherwise known from N.Z. Camptochaete arbuscula is also present in Hawaiʻi, and Weymouthia mollis is shared with Tahiti (Allen & Magill 2003).