- ≡ Dicranum introflexum Hedw., Sp. Musc. Frond. 147 (1801)
- = Dicranum leptocephalum Müll.Hal., Bot. Zeitung (Berlin) 9: 551 (1851)
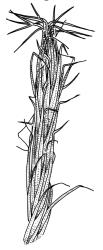
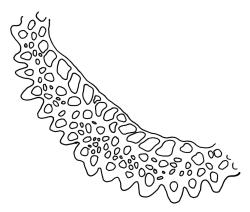
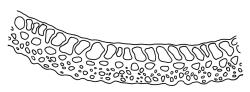
Plants yellow-green or rarely dark brown above, usually brown or ± black below, wider above in both ♂ and ♀ plants and appearing ± comose, forming dense turves on soil or decaying wood. Stems highly variable in height, c. 5–35 (–60) mm, mostly branched by innovation, in cross-section with a central strand, several layers (c. 5–7) of thick-walled cortical cells and no hyaloderm, beset below with smooth, orange-brown rhizoids. Leaves erect spreading and variably reflexed apically when moist, tightly appressed at base and with some leaves strongly squarrose-reflexed apically when dry, usually more crowded (± comose) near stem apices, often serially comose in fertile material, gradually subulate from an oblong base, strongly concave to subtubulose, mostly ending in a long hyaline, toothed, and usually squarrose-reflexed hair-point but occasionally concolourous at apex, not bordered above base, mostly 3.5–5 × c. 0.5(–0.8) mm (flattened); basal laminal cells thin-walled and hyaline, narrowly rectangular, variable in length but commonly c. 45–105 µm long, with a ± abrupt V-shaped transition to shorter, more rhombic cells above; upper laminal cells mostly short-rhombic or quadrate, rounded at corners, incrassate, not porose, some oblate and obliquely oriented (especially a short distance above the boundary with the basal laminal cells; further up the lamina frequently becoming more irregular to rhombic), mostly 12–24 × 6–12 µm; alar cells usually indistinct, not or weakly inflated, hyaline or pigmented; marginal cells at mid leaf more quadrate or oblate in several ill-defined rows, not forming a distinct border. Costa c. 290–380(–500) µm wide and c. 50–70% of the leaf base width, at mid leaf well defined and c. 210–280 µm wide, filling the upper subula and excurrent in a hyaline, toothed hair-point, which is usually squarrose when dry, lacking lateral spurs (unbranched); in abaxial surface (mid leaf or above) view appearing as ranks of short-rectangular to quadrate cells which alternate (some fine focusing required) with ranks of more elongate cells in the grooves; in adaxial view (mid leaf) the cells appearing uniformly elongate (mostly >150 µm long), ± thin-walled, and with oblique terminal walls; in cross-section (mid leaf) with a moderately distinct median layer of guide cells, a single layer of large (c. 8–14 µm in greater diam.), thin-walled, and exposed adaxial hyalocysts, and 3–5 abaxial layers of stereids (including cells of abaxial ridges), with numerous 1-celled abaxial ridges. Propagula usually absent.
Dioicous. Perichaetia clustered (to at least 10) in a terminal cluster, each giving rise to a single fruit; perichaetial leaves after fertilisation strongly sheathing the setae, longer and narrower than vegetative leaves. Perigonia aggregated in a terminal cluster, each surrounded by broadly ovate, concave, and pigmented bracts. Setae strongly cygneous, c. 8 mm; capsules ovoid, variably strumose, sulcate when dry, c. 1.8 mm; exothecial cells elongate and vermicular, with thick and strongly sinuous walls; stomata absent; annulus well-developed and apparently remaining attached to the fallen operculum; operculum long-rostrate from a conic base, c. 0.8 mm and half the capsule length. Peristome teeth orange and vertically striolate below, hyaline and baculate above, divided c. half-way to base. Calyptra fimbriate. Spores 12–14 µm, smooth.
Catcheside 1980, fig. 43; Beever 1986, fig. 2 (as C. catarractilis); Beever et al. 1992, fig. 23, b; Frahm 1987, fig. 12; Malcolm & Malcolm 2003, p. 12; Meagher & Fuhrer 2003, p. 107; Seppelt 2004, fig. 51.
In N.Z., the distinction between C. introflexus and C. bicolor can be difficult and the two species can grow intermixed. The two cannot always be differentiated in the field, although populations with abundantly piliferous and strongly reflexed leaf apices can assuredly be recognised as C. introflexus. Features useful for the separation of these two species are discussed under C. bicolor.
K; NI: N Auckland including offshore islands (TK, PK, HC, LB, RT), S Auckland, Gisborne, Hawke’s Bay, Taranaki, Wellington; SI: Nelson, Marlborough, Canterbury, Westland, Otago, Southland; St; Ch; Sol; A; Ant; C; M.
Austral. Tasmania*, mainland Australia*, Argentina*, Falkland Is.* Material which appears to be correctly named has been seen from Rapa and the Austral Is and Whittier (1976, p. 83) recorded it from the Society Is, New Caledonia, and New Guinea. Frahm (1987) described its range as "widespread throughout the southern hemisphere and introduced in western North America and Europe as an aggressive spreading species of open habitats". Gradstein & Sipman (1978, fig. 3) mapped the distribution on a world scale.
Forming turves of up to several square metres in extent on mineral or humic soils, duff, peat, rotten or burnt wood. Often abundant in disturbed situations, such as at roadsides, and following fire, but also occurs in a wide variety of undisturbed vegetation types including bogs, kānuka, and mānuka scrub, exotic plantations, and coastal dunes. Sometimes occurring on exposed roots or fence posts. C. introflexus is a weedy species of wide environmental tolerance occurring throughout N.Z. Accordingly, it co-occurs with a wide range of weedy species, including other species of Campylopus. From near sea level (Waikuku Flat, North Auckland L.D. ) to 1280 m (Mangawhero Falls on Mt Ruapehu, Wellington L.D.) on the North I., and to c. 1580 m (Arnaud Range, Nelson L.D.) on the South I.
In most instances C. introflexus is readily recognisable when dry by its conspicuous and strongly reflexed hyaline hair-points. Frequently the reflexed hair-points are visible only on comal leaves or, in sterile material, only in leaves at the stem apex. In Tasmania fertile shoots can be quite long and nearly the upper stem length cited above, and then the shoots take on the more club-shaped appearance (R.D. Seppelt, pers. comm., 18 Oct. 2017). The hair-points can rarely appear yellow in herbarium collections. Sterile material tends to be more elongate and have shoots much narrower (c. 0.6 mm diam. when dry) than plants bearing sex organs. The abrupt V-shaped transition from the thin-walled and hyaline basal laminal cells to upper laminal cells is also a characteristic feature, especially when viewed in combination with the short-rhombic, quadrate, or oblate and very compact upper laminal cells, which are rounded at the corners, incrassate, not porose, with some oblate and obliquely oriented. Male plants are often produced in abundance and are conspicuous when colonies are viewed from above. Unusually for the genus, this species, although dioicous, commonly produces abundant capsules.
The abaxial costal ridges in mid leaf cross-sections are usually very distinct but only a single cell in height; these likewise aid in recognition. In highly magnified abaxial surface view the ridges appear as ranks of short-rectangular or quadrate cells, which alternate (some fine focusing required) with ranks of elongate cells in the grooves. The abaxial ridges, in well-developed material, can thus often be visualised in surface view. Catcheside’s (1980) fig. 43, f, is informative and accurate. In sections from near the leaf insertion, the ridges are mostly completely lacking (cf. Catcheside, 1980, fig. 43, e), while a section from just below the hair-point base usually differs little from that at mid leaf. At mid leaf the adaxial hyalocysts are conspicuous in cross-section.
Sometimes otherwise representative plants (e.g., K.W. Allison 613 from Kaingaroa Plains, CHR 545967) give rise to propagulae in which the reduced leaves are apically concolourous and ± cucullate, and usually have poorly developed abaxial costal ridges. According to J.E. Beever (pers. comm., 18 Aug. 2017), the leaves of the propagula of C. introflexus are shorter and more or less erect compared to those of C. clavatus, and this feature can be used to distinguish the two in the field.
There is some confusion around the typification and type locality of Dicranum introflexum Hedw. Hedwig (1801) gave the type locality as "Insularum meridionalium incola" and cited no collector. Richards (1963) seems to have been the first modern author to attempt to identify a potential type and to clarify the application of the name C. introflexus to British and western European collections. However, Richards did cite an earlier study by Giacomini (which has not been seen). Most significantly in an Australasian context, Richards provided notes on potential type material of Dicranum introflexum Hedw. in the Hedwig-Schwägrichen herbarium at Geneva. He based his notes on photographs of the three herbarium sheets in that herbarium, filed under the name C. introflexus, rather than examination of the actual specimens. He cited "one of the specimens on sheet 1(labelled ‘A’) [as] "certainly C. introflexus (Hedw.) Brid. as understood by Giocomini". There seems no doubt that this material was collected in Australia ("Nova Holl."). Richards’s notes strongly suggest (but do not specifically state) that this material was collected by Sieber. His notes have been cited by subsequent authors (e.g., Gradstein & Sipman 1978; Frahm 1987) and the particular specimen he mentioned has been considered type material, if not an actual lectotype. There seems to be no potential type material of Dicranum introflexum Hedw. in Australasian herbaria. The consensus concerning the type of D. introflexum is not discussed further in this treatment; the controversy concerning its occurrence in Britain and Europe is discussed very briefly below.
Dixon (1923, p. 89) listed only one heterotypic synonym of C. introflexus that is based on a N.Z. type. Definite type material of Dicranum leptocephalum Müll.Hal. has not been located but there seems no reason to question Dixon’s judgement. Material named as D. leptocephalum in Beckett’s herbarium (CHR 642104) that is either a portion of Mossman’s type collection or collected by R. Helms near Greymouth is correctly referred to C. introflexus.
Campylopus novae-zealandiae E.B.Bartram & Dixon in Dixon & E.B. Bartram [Bot. Not. 1937: 72, 1937] is a probable synonym here. The Southland L.D. type was collected at Bluff between Invercargill and Winton by S. Berggren in 1874. An isotype (WELT M009420!) is extremely sparse, consisting of four stems each less than 15 mm long. The leaves have a short base abruptly tapered to a long and slender acumen that is entire except for moderate toothing at its extreme apex. On the basis of both its areolation and costal morphology it is best to tentatively refer this material to C. introflexus, despite its lack of squarrose hyaline leaf tips. Examination of more ample type material would probably confirm this placement.
The Australian Campylopus pudicus (Hornsch.) A.Jaeger was accepted as a N.Z. species by Dixon (1923, p. 90) but the single Southland L.D. collection made by J. Meiklejohn (WELT M 001238) and cited by Dixon is C. introflexus. I have also seen one Berggren collection from Taupō (CHR 544543) named (by Sainsbury?) as C. pudicus and consider it referable to C. introflexus.
Campylopus catarractilis (Müll.Hal.) Paris [Index Bryol. Suppl. I, 90 (1900)], a South African species, was recorded from littoral rocks on the Poor Knights Is by Beever (1986, see also Beever et al. 1992 and Fife 1995), based on determinations by J.-P. Frahm. However, re-examination (including independently by J.E. Beever) of the relevant Poor Knights collections shows that this material is a depauperate, turf-forming, coastal seepage form of C. introflexus, with reduced abaxial costal ridges but no other significant microscopic differences from this species. In the most ample collection referred by Frahm to C. catarractilis (J.E.Beever 30-18c, AK 206141), the majority of the plants have blunt to slightly rounded, scabrid, and concolorous leaf apices, some of which are weakly cucullate. However, plants are also present in which the upper leaves have a short, reflexed, hyaline hair-point. Plants with/without hair-points are indistinguishable in their mid laminal cell shape and dimensions, costal cross-sections, and the nature of their basal laminal cells, and in all these features are representative of C. introflexus. Similar material, with mostly concolourous but acute leaf apices, has been seen from the Hen and Chicken Islands (N. Auckland L.D.) and from the Black Rocks (Bay of Islands, N. Auckland L.D.), collected by L.B. Moore and Jessica Beever, respectively.
There is a large literature concerning the occurrence of C. introflexus in Britain and Europe, where it is considered an invasive adventive. Richards (1963) seems to have been the first to try to clarify this confusion in a British context. According to Smith (2004), the first collection of C. introflexus in Britain was made in 1941 and the first in Ireland in 1942. Confusingly, however, the name C. introflexus was applied to British material much earlier by Dixon & Jameson (1896), as well as in other 19th century British/European literature. The confusion seems due to the difficulty in distinguishing the adventive C. introflexus from the indigenous C. pilifer Brid. (=C. polytrichoides De Not.). This problem is beyond the ambit of this Flora; it has been discussed by several authors, including Frahm (1972) and Gradstein & Sipman (1978).